Jeffrey Alston

Jeffrey Alston
Physical and electrochemical interactions within hybrid nanocomposites of ruthenium coordination complexes and single-walled carbon nanotubes
Abstract:
The research presented in this dissertation is a study of the interaction of ruthenium coordination complexes with single-walled carbon nanotubes (SWCNTs), a pursuit ultimately leading to the development of composite SWCNT materials. The work comprising this dissertation includes three major accomplishments: the synthesis and characterization of two new dinuclear ruthenium coordination complexes, the development of isothermal titration calorimetry (ITC) to thermodynamically quantify interactions with SWCNTs, and the fabrication and characterization of ruthenium complex—SWCNT hybrid nanocomposite electrodes.
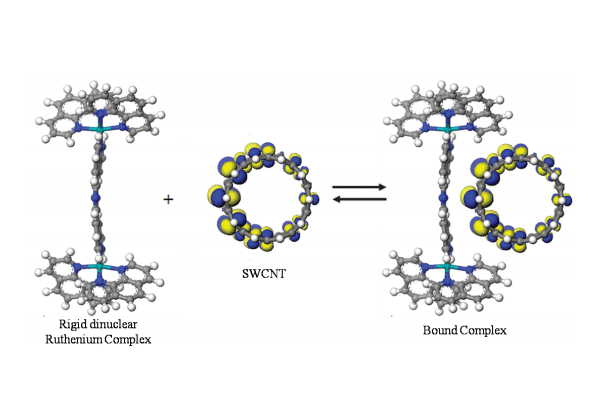
The work leading to these major accomplishments is inspired by the goal of attaining control over assembly of nanoscale building blocks, i.e. SWCNTs. The first step towards this goal is the development of appropriate molecules that can nondestructively link two SWCNTs together without damaging the physical structure of the tube. [Cl(trpy)Ru(tpphz)Ru(trpy)Cl](PF6) 2 and [(phen)2Ru(tpphz)Ru(trpy)Cl](PF6)3 are the two ruthenium dimer molecules synthesized and discussed herein. They possess a rigid nanoscale pocket that contains conjugated π-electron density capable of interacting with the walls of SWCNTs. During the work to synthesize these complexes significant improvements were made to synthetic procedures to produce important precursors. The synthesis of the two complexes and the new synthetic procedures were novel.
The second step required the development of a new tool (ITC) to study the interaction thermodynamics of dispersions of SWCNTs. ITC is a well established tool to measure binding thermodynamics of biological proteins and enzymes. Based on the analogy that can drawn between SWCNTs in solution and proteins, I developed ITC methods and protocols for measuring interactions of solvents with SWCNTs as well as the binding of the ruthenium dimer complexes with SWCNTs. I have established that ITC can be an important nanoscale science and materials development tool which can provide detailed insight into the thermodynamic interactions of nanomaterials in solution.
I combined SWCNTs and ruthenium complexes, and developed procedures to fabricate nanocomposite films. The films produced by our method improve on previously reported techniques by avoiding surfactants and binders which retard the properties of SWCNT films. I was able to transfer these films to various substrates and they were shown to have enhanced capacitance versus pristine SWCNT films when used as an electrode in an electrochemical cell. Augmenting SWCNT electrodes in this way has not been reported and the technique is a promising vehicle for photo-induced charge transfer as well as cheaper and lighter capacitor devices.
Sponsoring Chair: Dr. Jordan Poler
Committee: Dr. Steve Patterson, Dr. Markus Etzkorn, Dr. Thomas Schmedake, Dr. Maciej Noras